|
DILUTION PLATING Purpose This procedure is used to identify the number of viable micro-organisms in a fixed amount of a liquid. It can also be fairly easily modified to give results with solid substances, e.g. Macerated foods. Background Serial dilution involves repeatedly mixing known amounts of source culture with (sterilised) liquid. 1 ml added to 9 ml gives a 10-fold dilution; 1 ml added to 99ml gives a 100-fold dilution. When fixed amounts of this dilution series are mixed with an appropriate agar and incubated, then different numbers of colonies will be obtained. Study 5 Serial Dilution method flashcards from Mandy S. On StudyBlue. Microbiology & Molecular Biology. Advantages of serial dilution-agar plate; 1. The dilutions cover the range from 1/2 to 1/100 unevenly. In fact, the 1/2 vs. 1/3 dilutions differ by only 1.5-fold in concentration, while the 1/10 vs. 1/100 dilutions differ by ten-fold. If you are going to measure results for four dilutions, it is a waste of time and materials to make two of them almost the same. Serial dilution only requires the measurement of a volume of the solution of known concentration one time. Advantages of “Serial Dilutions. By working back from an easily counted plate and using the appropriate dilution factor, the number of micro-organisms in the original source culture can be calculated. Procedure For this exercise, yeast suspensions, ('fresh' or 'stale') milk, or water may be used. Lay out and label the tubes and (empty) Petri dishes as shown in the diagram below. Each member of the team can take it in turns to perform the repeated sections below, and prompt others as required. Flame and loosen the lids of tubes 0 and 1. Using a sterile pipette HANDLED ASEPTICALLY transfer 1 ml of liquid from tube 0 to plate 0, and USING THE SAME PIPETTE, transfer 1 ml of liquid from the source culture (tube 0) to tube 1. 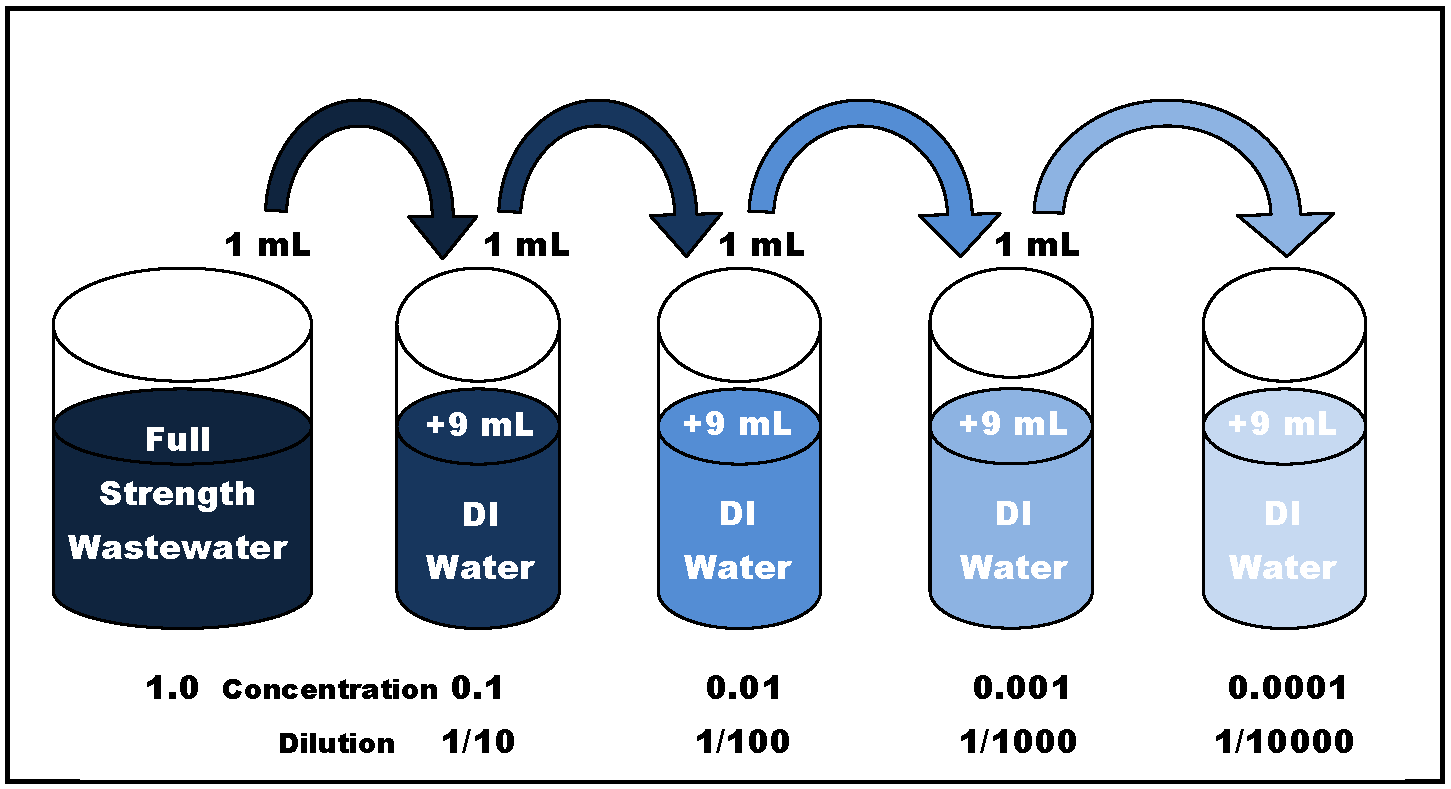
Then: DISCARD THE PIPETTE. Flame the edge of tube 1. Seal and mix the contents gently. Repeat the process with the next tube and plate: Flame and loosen the lids of tubes 1 and 2. Transfer 1 ml of liquid from tube 1 to plate -1, and also into tube 2. Serial DilutionDISCARD THIS PIPETTE. Flame the edge of tube 2. Seal and mix the contents gently. Repeat the same steps, 5 or 6 times, moving along the chain as shown in the flow chart below. At the end of this process: Take a bottle of sterilised agar from the 45 °C waterbath, where it has been kept just above setting temperature. Dry the outside of the bottle, and flame the top and neck area. Batch file echo prompt. Then WORK QUICKLY AND ASEPTICALLY: Opening each Petri dish lid only slightly, pour nutrient agar into the dilution liquid already in the Petri dish, until it covers about two thirds of the area - although this is not critical.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
